Vascular Evaluation in Patients with Lower Extremity Ulceration

Blogs

Blogs
Vascular Evaluation in Patients with Lower Extremity Ulceration
INTRODUCTION
Over 30% of lower extremity ulcers and nonhealing wounds are not fully resolved at six months even though patients have received "best care available" and spent 10's of billions of dollars in direct care costs.1-4 Nearly 15% of Medicare beneficiaries have lower extremity ulcers with a conservative estimated annual cost exceeding $30 billion dollars.5 Compounding this is the rising prevalence (currently estimated at 1-2%) of leg ulcers as people are living longer.6 Despite an abundance of studies and guidelines, there is significant variation in recommendations, which are often contradictory, causing confusion and raising barriers to patients receiving appropriate care.
Deficiencies in the management of leg ulcers are common and are due to incorrect diagnosis and treatment.7-9 Inappropriate treatment may cause deterioration of the wound, delayed wound healing, and harm the patient. Barriers to healing include lack of consensus, interprofessional rivalry, no standardization, care delivery structure, limited resources, and educational gaps.10-11 The delivery of health care services is in constant flux, which is detrimental to interdisciplinary teamwork, professional relationships, and a collaborative environment.
This brief addresses the underappreciated role of arterial and venous disease in patients that have lower extremity ulceration. Significant knowledge gaps are addressed, including the often-misunderstood importance of physical exam findings and the interpretation of physiologic and ultrasound testing.
amputation lottery
A "Pathway to Amputation" characterizes the care of patients with limb-threatening ischemia in the United States.12 A 2012 study of 20,464 Medicare patients with limb ischemia showed that 71% of patients had no attempt at revascularization and 54% did not even have an angiogram before major amputation.13
Numerous studies have shown that the probability of undergoing a major amputation depends on who you are and where you live: the "Amputation Lottery." 14,15 In addition to the lost quality of life for the patient, primary amputation is costly, exceeding the cost of endovascular revascularization by almost $10,000.14
The employment of primary care physicians by health care "systems" aggravates patients' access to receiving the best care.16 Hospitals are limiting referrals to outside specialist care: "Leakage versus Keepage." 17 This restriction of physician and patient choice has led to legal action and litigation.18,19 In 2012, a jury awarded $1.23 million because of untimely referral.20
KNOWLEDGE GAPS
Nonhealing ulcers and wounds are not a disease but a symptom of an underlying condition.21 Successful treatment depends on an accurate diagnosis of the underlying cause. The vast majority, over 85%, of lower extremity wounds have an arterial, venous, or mixed arterial and venous disease as a primary cause.22-26
Clinical diagnosis based on ulcer anatomical location, characteristics, and morphology have received much emphasis.23,27 While useful, this approach can cause tunnel vision and result in patients not receiving appropriate, timely care. Old maxims, such as all plantar wounds are neuropathic, or venous wounds only occur in the setting of varicose veins, are wrong (Figure 1).
When evaluating a patient with a lower extremity wound, the primary task is to rule out an underlying correctable contributing etiology. This requires in-depth knowledge and understanding of the utility of clinical evaluation and diagnostic testing to include or exclude arterial and venous disease.28-30 There are many physiologic and imaging tools to identify patients with arterial and venous disease. Most of these tests are "good tests" but not "great tests." They must be interpreted with an understanding of the sensitivity, specificity, and disease prevalence.31,32 In short, the question is: "what is the chance that the clinical condition will be present or absent in the context of a positive or negative test?" 30
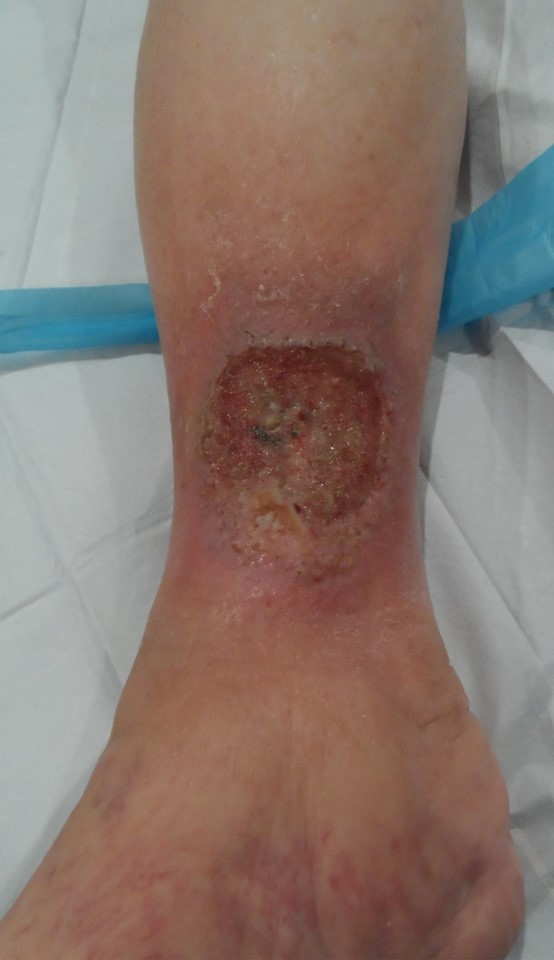
Figure 1. Venous ulceration without other signs of venous disease.
ARTERIAL DISEASE
One of the most critical steps in evaluating a patient with a lower extremity ulcer is to exclude ischemia as a potential contributor to the wound and determine if revascularization is required. Appallingly, in the United States, almost 50% of patients who undergo a significant lower extremity amputation have not had an appropriate arterial evaluation prior.12,13,33 An in-depth discussion of the nuances of evaluating lower extremity arterial perfusion in the setting of chronic limb-threatening ischemia (CLTI) is the subject of another VIE Brief "Evaluation of Foot Perfusion." The essentials of arterial testing are covered here.
At the bedside, our primary tools include identifying risk factors for peripheral arterial disease (PAD) and physical examination findings. Important risk factors include (but are not limited to) smoking, diabetes, age, cardiovascular, or cerebrovascular disease. Physical exam findings include pulses, capillary refill, trophic changes (loss of hair, dry, scaly skin), and ulcer characteristics. These tools are useful for identifying patients with PAD – "rule in."
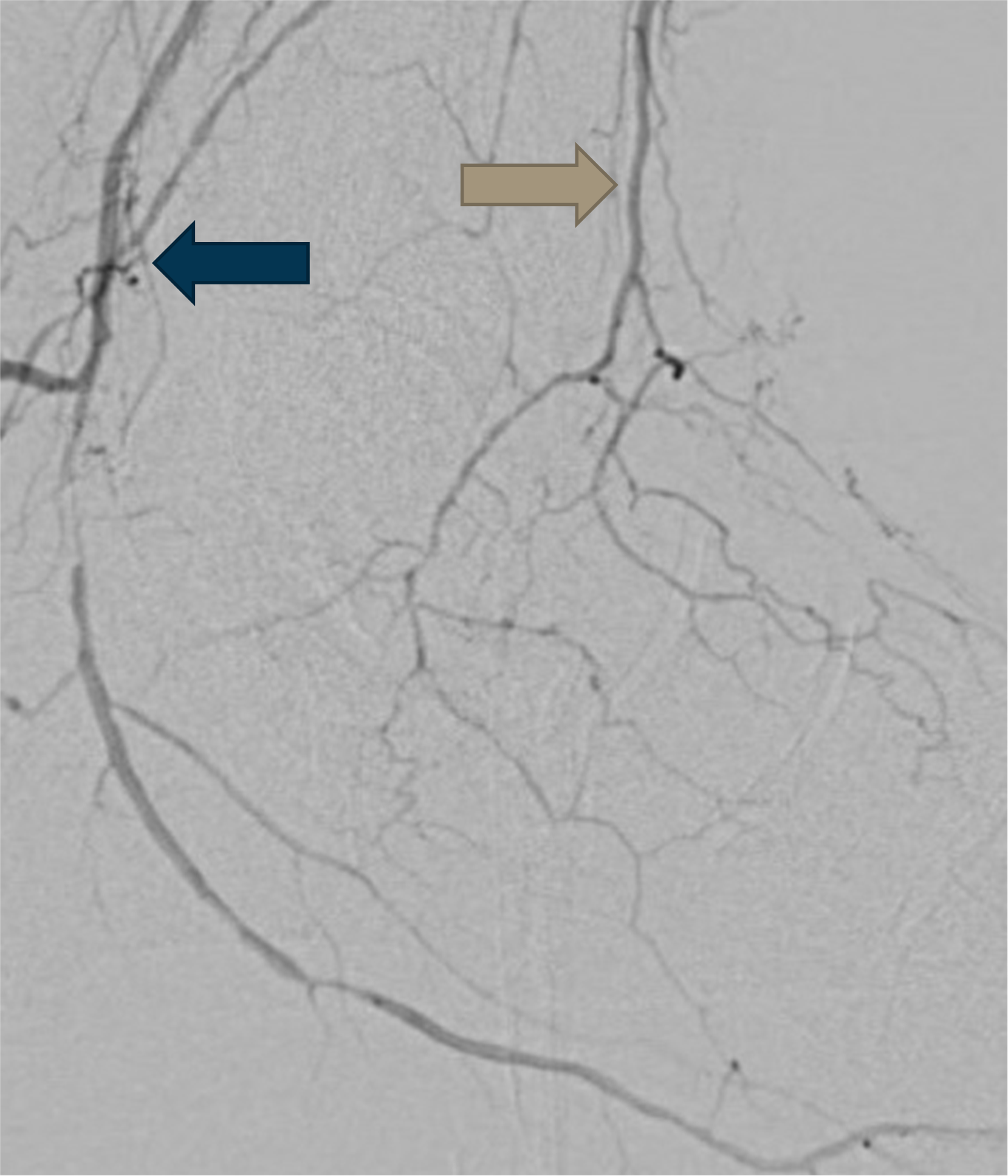
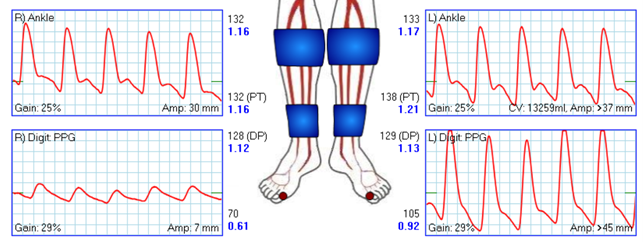
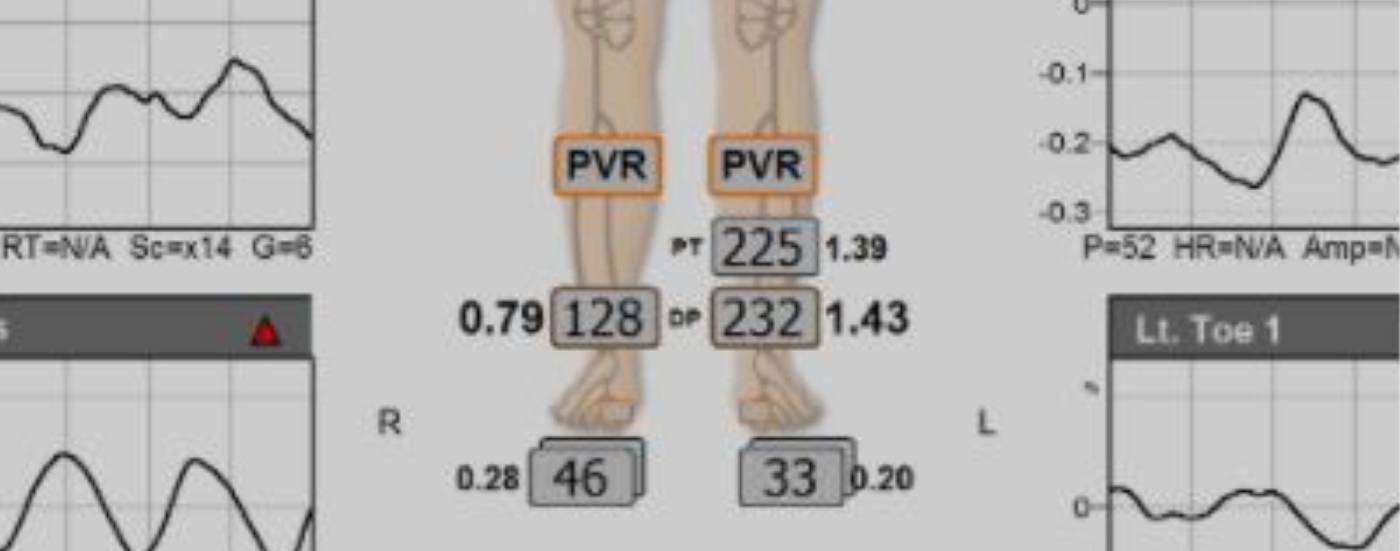
Figure 2. Normal ABIs with abnormal TBI and TP on the right.
Figure 3. Angiogram showing occlusion of the common plantar artery and dorsalis pedis artery. The PTA and ATA pulses were palpable.
However, these clinical tools are not useful to "rule out" significant PAD.34-36 More specifically, the clinical examination for identifying ischemia has a "poor negative predictive value and should not be used in isolation to triage patients with nonhealing ulcers".37 The patient shown In Figure 3 had palpable pulses and was wrongly assumed not to have significant PAD.
The presence or absence of PAD must be determined using objective testing. Objective tests for PAD are broadly classified into two categories: physiologic and anatomical. Physiologic testing includes ankle-brachial indices (ABI), toe brachial indices (TBI), toe systolic pressures (TSP) segmental pressures, skin perfusion pressures, transcutaneous oxygen tension, pulse volume recordings, Doppler waveforms, and other newer, not well-validated modalities. Non-invasive anatomical imaging includes an arterial ultrasound (US), CT angiography, and MR angiography. Physiologic testing should always be utilized before anatomical imaging.38
Frustratingly, there are difficulties and limitations with all currently available physiologic evaluation modalities. Surprisingly, the ABI, widely considered the gold standard for detecting PAD, has limited negative predictive value in the setting of CLTI, being falsely reassuring and falsely negative in 30% or even 40% of patients.39-43 An ABI should not be used in isolation when evaluating for CLTI as it is not an accurate indicator of pedal perfusion.42,44
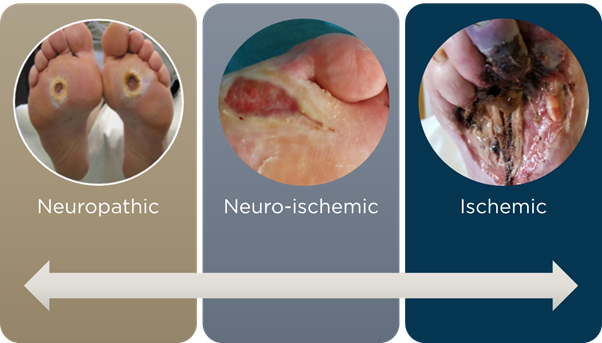
Figure 4. Spectrum of foot ulceration.
Measuring blood flow at the level of the toes is mandatory in patients with CLTI to improve the detection of the presence of significant ischemia (Figure 2).45-48 Many nuances need to be considered when evaluating pedal blood flow.49,50 TBIs and TSPs should not be confused with one another. TBIs should be thought of as screening test, while TSPs should be thought of as a diagnostic test.49 Alternatively, skin perfusion pressures (SPP), although not as well validated, are another option for vascular testing in patients with CLTI.51
Diagnosing PAD is not always straightforward.52 A pragmatic approach, starting with a detailed history and physical exam, followed by testing TBI/TSP or SPP, is a cost-effective strategy.53 Clinicians must recognize that foot ulcers occur on a spectrum from neuropathic to neuro-ischemic to ischemic Figure 4.54 Finally, before any amputation, referral to a critical limb expert for catheter angiography should be mandatory.55
VENOUS DISEASE
Venous ulceration is hugely distressing for patients. It significantly affects their quality of life, causing about 40% more days of lost employment than any other disease.56,57 Our knowledge of pathophysiology, hemodynamics, diagnostic imaging, and treatment of chronic venous insufficiency (CVI) has expanded dramatically, particularly in the last decade. Venous pathology develops from valvular incompetence, obstruction, and muscle pump dysfunction causing venous hypertension.58
Like PAD, the clinical findings (varicose veins, hyperpigmentation, stasis dermatitis, atrophie blanche, lipodermatosclerosis) are useful to "rule in" CVI. There are no studies on the utility of the lack of these findings to "rule out" significant venous disease. Unfortunately, there is the all-too-common misconception that the absence of these clinical findings implies no significant venous disease. The lack of varicose veins or leg swelling does not confer that a patient does not have substantial venous disease.59 This can only be determined by objective testing.
Non-vascular specialists poorly understand testing for CVI.60 This is due to limited education during medical training, rapidly evolving knowledge, and various tests available.
Not all venous US studies are the same. Most (all) clinicians are familiar with ordering a venous US to evaluate for deep venous thrombosis (DVT). This examination has one purpose - to determine if a patient has or does not have DVT. Unfortunately, even this widely used test is inconsistently performed and can have a higher false-negative rate than expected.61 Physicians must recognize that a DVT US study does not evaluate for venous valvular function or venous obstruction.
Venous incompetence US is the primary diagnostic tool for evaluating venous valve function in both the lower extremity's superficial and deep venous systems.62,63 The adequate performance of a venous incompetence US requires specially trained and experienced US technologists. The supervision and interpretation of these US studies must be performed by a specialist with a deep understanding of the complexities of lower extremity venous disease.64
There are two categories of venous obstruction, inflow, and outflow. Inflow disease is an obstruction in the deep veins (common femoral, femoral, and popliteal veins) of the leg and outflow disease as obstruction of the iliac veins or the inferior vena cava in the pelvis or abdomen.
The importance of venous outflow obstruction in CVI is becoming better understood.65-67 Non-invasive diagnostic imaging, including venous US, CT venography, and MR venography can be useful but lack well-validated parameters for determining outflow disease. Invasive imaging with intravascular ultrasound (IVUS) is the best tool for identifying patients with significant venous outflow compression.68
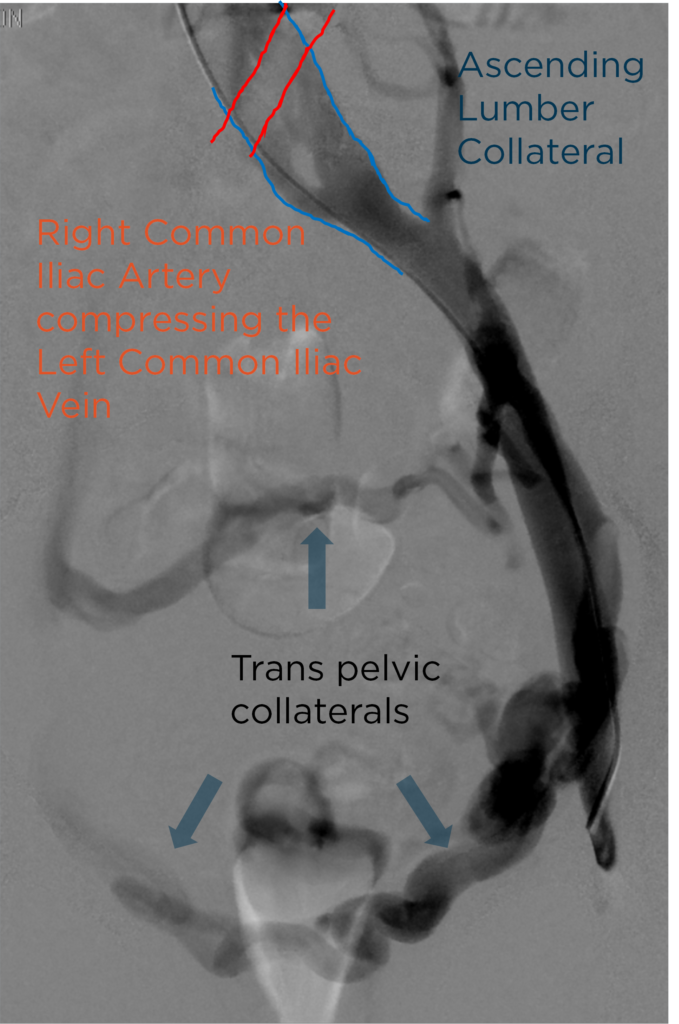
Figure 5. Venography showing severe compression of the left common iliac vein by the right common iliac artery.
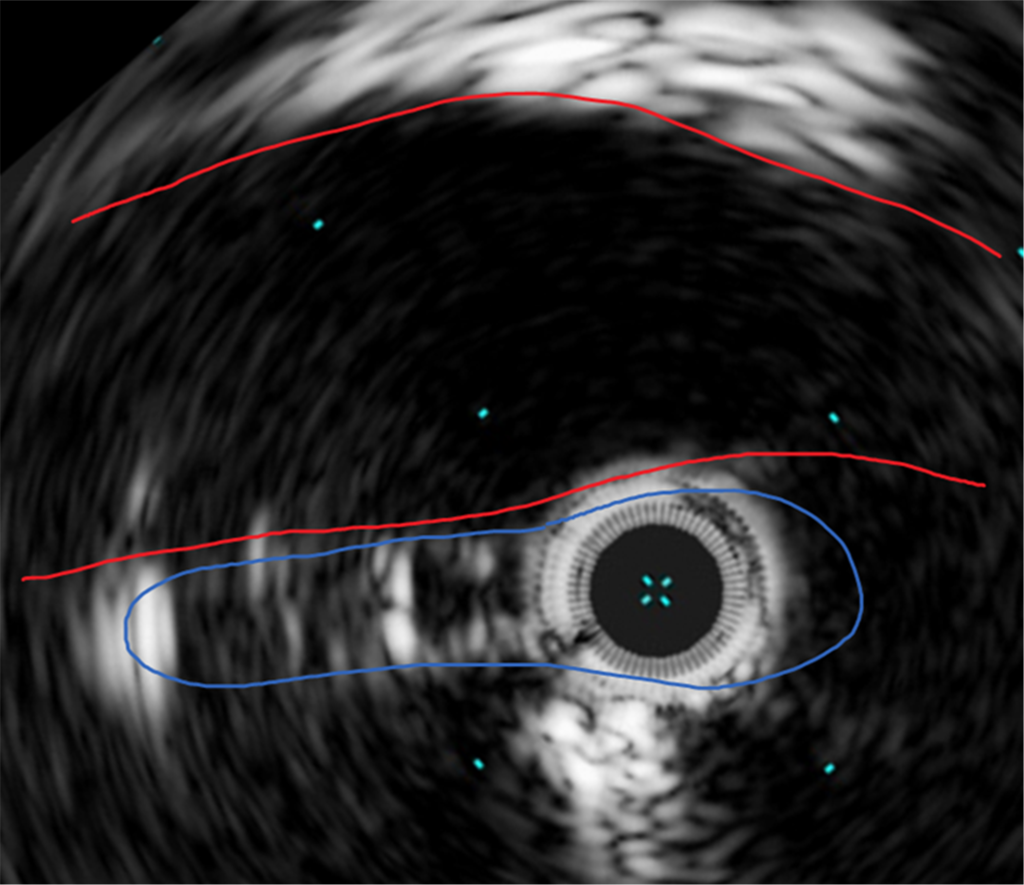
Figure 6. Intravascular ultrasound showing severe compression of the left common iliac vein (blue) by the right common iliac artery (red).
Inflow venous obstruction primarily occurs after lower extremity DVT and is one of the leading causes of post-thrombotic syndrome (PTS). Evaluation of the lower extremity deep venous system and potential therapies is an area of intense research.69,70
Finally, all patients with a suspected venous ulcer should have the ankle-brachial index measured at the time of initial presentation.71
CONCLUSION
Lower extremity wounds are complex and challenging for patients, families, and healthcare providers. Most lower extremity ulcers are secondary to either arterial or venous problems. A comprehensive history and physical assessment followed by appropriate imaging are essential to identifying, diagnosing, and developing care plans.
Dedicated vascular specialists should be involved in the care of all lower extremity ulcers and wounds to determine or exclude the presence of significant arterial or venous disease.7,72 Continuous advances in endovascular therapy from pedal artery intervention to arterialization of the venous system are improving outcomes and limb preservation rates (Figure 7).
Rapid referral and treatment of the underlying problem improves wound healing rates, reduces patient morbidity, and enhances life quality.4,73-75
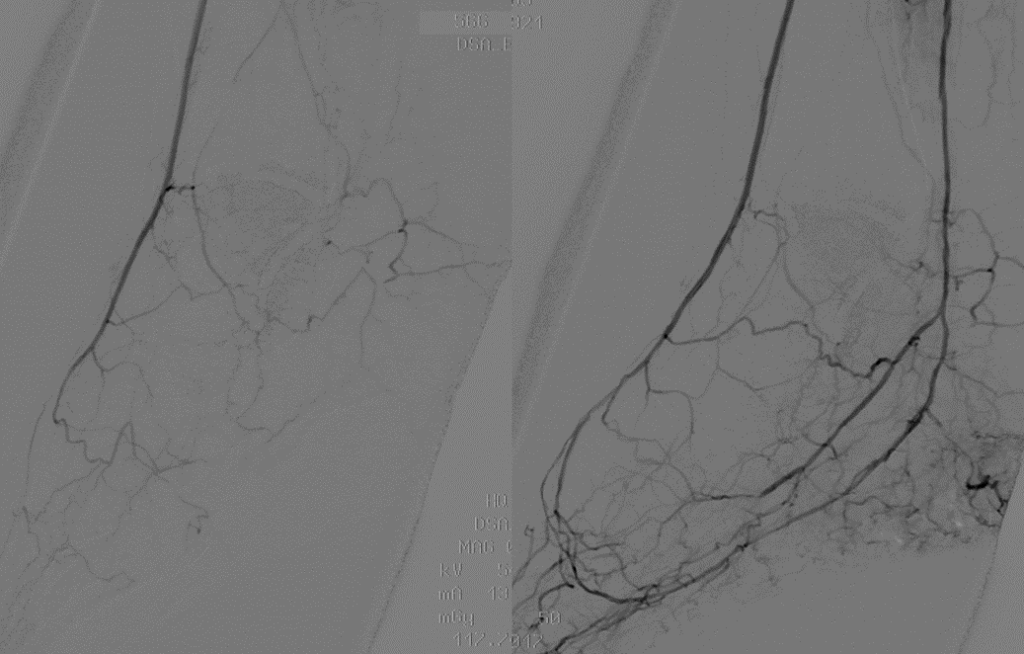
Figure 7. Endovascular reconstruction of the pedal loop and plantar arteries.

